According to a study by PWC (2011), the birth of the biotechnology industry dates back to 1980, when the US Supreme Court, allowed man-made, genetically engineered microorganisms to be patented. The OECD defines biotechnology as “the application of science and technology to living organisms to alter living or non-living materials for the production of knowledge, goods and services“. The last twelve months revenue generated by the biotechnology industry as of September 2016 was estimated at US$104 billion while the FDA approved 45 new drugs in 2015, the largest number since 1996, according to FierceBiotech.
A unique business model
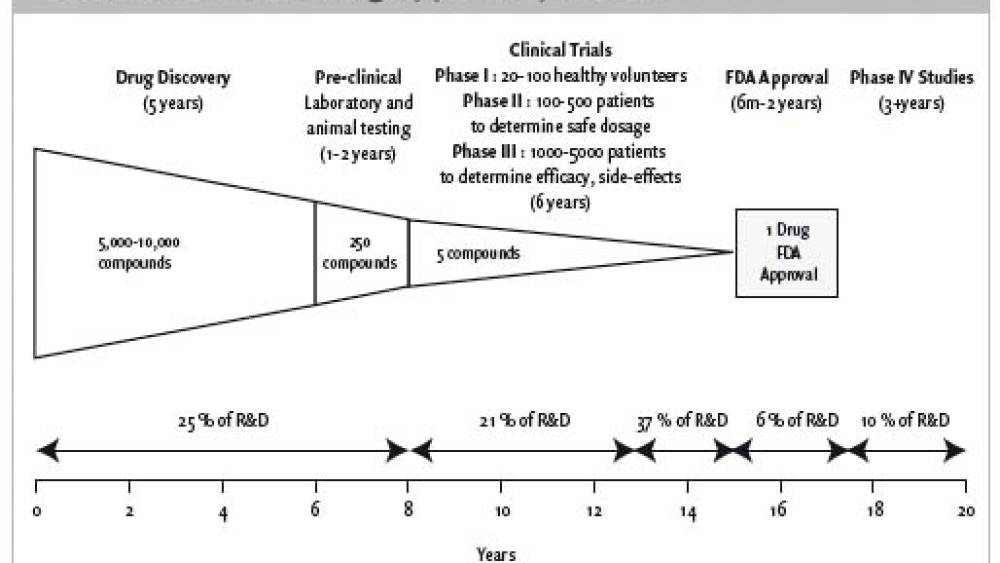
Biotechs have a very unique business model as they have to engage in major R&D whose costs can only be recovered when new products enter the marketplace, which can take 10-20 years. There is also high uncertainty underlying the success of a new drug and the highest research and development (R&D) spending corresponds to the development phases which have the lowest probability of success, therefore biotechs do not make a profit for many years, do not pay dividends, but show signs of growth. An approval to market a drug is a key event in a biotech’s life : before any new drug can be introduced to the market, it has to be approved by successfully passing 3 different phases, whereby the drug is tested for toxicity and effectiveness on healthy and diseased volunteers. A drug approval gives birth to likely revenue generation after years of spending, a potential switch to profitability, credibility and hopes for further drug approvals.
Biotechs’ financing
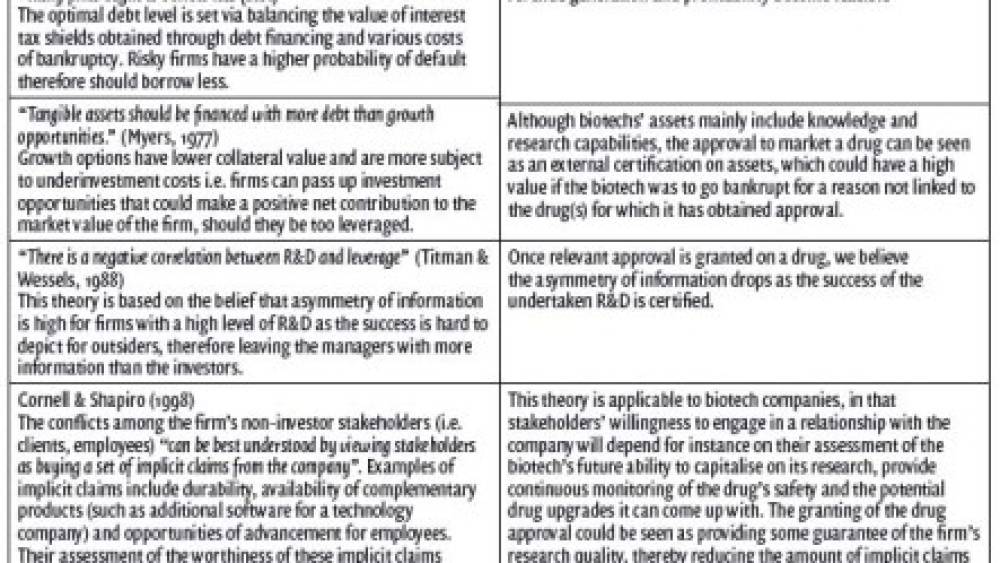
Early biotechs’ financing mainly include venture capital -which can be staged in order to raise capital in the future at a higher valuation- as well as R&D alliances, and, given investors are unlikely to commit their funds for 10-20 years, common exits are usually limited to IPOs and/or an acquisition by a larger firm (Booth, 2009). However, once a biotech has received a drug approval, one might wonder whether the financing of very early stages can still be appropriate. The review of theories on high-growth companies’ capital structure suggests that they do not apply to a biotech that has obtained one or more drug approvals (Chart 1).
Liu (2000)’s findings on the link between debt capacity and knowledge capital of US biotech firms - as measured by the number of i) patents applied for, ii) claims each patent has, and iii) subsequent citations since patent was approved - suggest that considering the successful output of research and development, as opposed to measuring the amount of R&D seems to yield different results, when applied to biotechnology companies. Liu’s findings, therefore, have important implications for the specification of the industry’s intangible assets the intangible assets of biotechnology companies have a unique specification and general theories on high-growth companies’ capital structure do not seem to be applicable. We hypothesised that biotechnologies that have one or more drugs approved finance themselves with more debt than the biotech companies, which haven’t yet obtained approval on a drug by the FDA.
Regression analysis
In order to determine the influence of having one or more drugs approved on biotechs’ capital structure, we ran a multiple regression on firms listed on the Nasdaq Biotech Index (NBI) between 2010 and 2015. We selected this specific time frame, as we believe the pre-financial crisis context differed from the post financial crisis market conditions in terms of investors’ appetite for risky debt. The biotechs’ financial data on Thomson One Banker while information on drugs approved by the FDA were found on Centerwatch.
Voir equation 1
We first ran a linear regression with all the control variables deemed significant by the literature on the study of capital structures, namely firm uniqueness (price to book ratio), size (natural logarithm of sales) and profitability (operating income as a % of sales). Asset tangibility is also a commonly used control variable however we could not find data for this. Uniqueness and profitability did not prove to be significant in the linear regression, most likely because the price to book ratios of the sample are very heterogeneous (the market value of a firm is very sensitive to any announcement) and since many biotechs in the sample did not have a drug approved by the FDA at the time of this analysis, their profitability was most likely negative and may have hindered the significance of the variable.
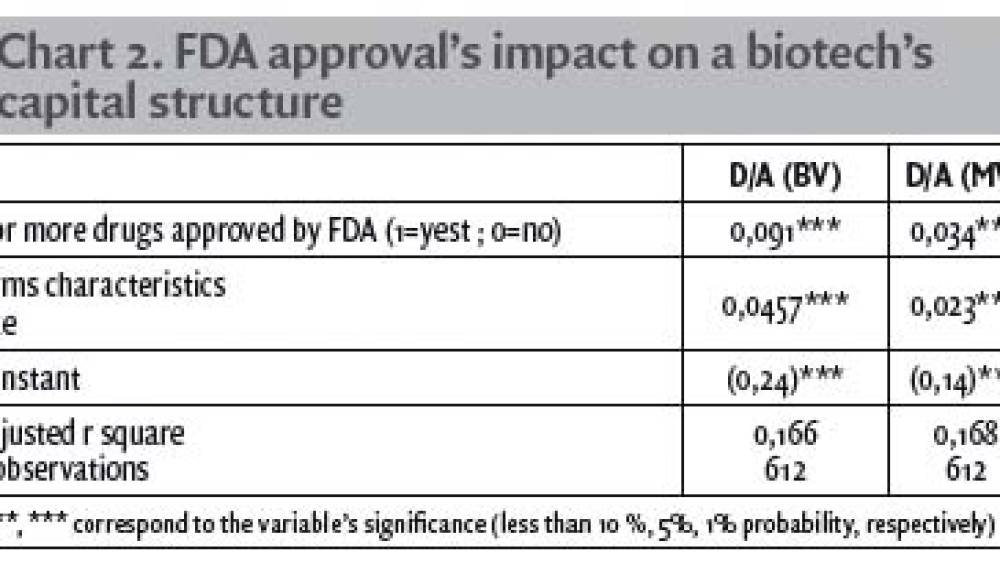
The FDA approval has a significant impact on a biotech's capital structure
The impact of the FDA approval has a significant impact on leverage: +9.1% on the book value leverage and +3.4% on the market value leverage. This is broadly in line with Liu’s model, which yielded a 0.02 coefficient for both leverages. This finding implies that size is relevant to the capital structure, as large firms are less subject to bankruptcy thanks to their diversity and establishment. Based on our empirical findings, we argue that the drug approval of a biotech by the FDA has a significantly positive impact on the firm’s leverage, due to the change in status that this event represents for the firm. These findings show that the FDA approval drastically modifies the status of the biotech firm, and that the specification of intangible assets differs among industries, given that, in the biotechnology industry, the FDA approval seems to provide a genuine certification of assets and bring comfort to lenders.
This finding can contribute to helping biotechs’ executives anticipate the change in the market’s perception on the firm and the potential new financing opportunities made available upon reaching drug approval. Potential biases of our empirical findings include that our hypothesis and methodology only deal with the United States, in the light of our willingness to focus on the FDA approval and on NBI listings. Although the United States is the main market for biotechnologies and the FDA is believed to have the strictest drug approval process, our model does not take into account the fact that a drug may have received prior approval from a different jurisdiction; therefore, our model may have failed to consider the debt raised by biotechnologies in regards to a drug approval they have obtained in a different jurisdiction, should they have not been approved yet by the FDA at the time of this study.